
The table below lists density of different metals including common metals and less common metals in g/cm3, kg/m3, lb/in3, lb/ft3. Briefly explain why chemically similar alkali metal chlorides, NaCl and CsCl, have different crystalline structures, while. The Pythagorean theorem is used to determine the edge length of. Common & Less Common Metal Density Chart / Table Solution Summary: The author explains that iridium forms a face-centered cubic lattice, and an atom is 271.4 pm. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is 10.49 g/cm3, for more metals, please view the metal density chart and table below. Use this information to calculate the radius of an atom of the element. The very dense metal iridium has a face-centered cubic unit cell and a density of 22.56 g/ cm3. Therefore, atomic radius of Iridium atom 1. Use this information to calculate the radius of an atom of the element. Given that Iridium crystallizes in a face-centered cubic unit cell. (Figure 1) Express your answer using four significant figures. Use Avogadros number to calculate the atomic mass of iridium. The edge length of the unit cell was found by x-ray diffraction to be 383.9 pm. One atomic mass unit is equal to 1.66 x 10 -24 grams. 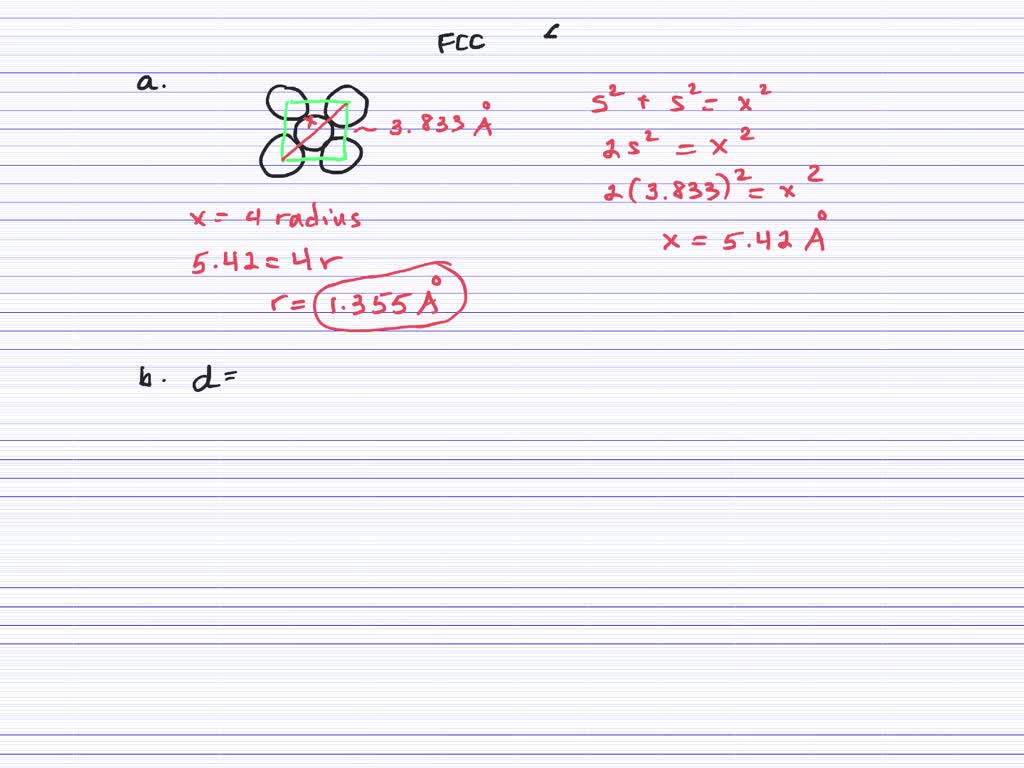
The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. To find the density of the iridium metal, we need to find the mass of atoms per unit cell. Part B Calculate the density of iridium metal. Iridium metal crystallizes in a face-centered cubic structure. Note that each element may contain more isotopes. For example, the most dense metal is Osmium (Os), with a density of 22.59 g/cm3, which is 42 times that of the least dense metal Lithium (0.534g/cm3). The density of iridium metal is 22.67 g/cm³. Iridium's name is derived from the Greek goddess Iris, personification of the rainbow, on account of the striking and diverse colors of its salts.Density of Metals – List of Metals by Densityĭifferent metals have different densities, and the density difference between some metals is huge. Iridium is found as an uncombined element and in iridium- osmium alloys. 1 Equilibrium diagram of the iridium-platinum system Platinum Metals Rev., 1960, 4, (1) 19. high density made the alioys very suitable for the production of standard weights. It will not react with any acid and can only be attacked by certain molten salts, such as molten sodium chloride. at the French Mint to determine the suit- ability of iridium-platinum alloys for coinage. It is the most corrosion resistant metal known and is the second-densest element (after osmium). Iridium is a member of the platinum group of metals. In its elemental form, Iridium has a silvery white appearance. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Iridium was discovered and first isolated by Smithson Tennant in 1803. Calculate the radius of an iridium atom given that Ir has an FCC crystal structure, a density of 22.4 g/cm3, and an atomic weight of 192.2 g/mol. The iridium atom has a radius of 136 pm and a Van der Waals radius of 202 pm. The number of electrons in each of iridium's shells is and its electron configuration is 4f 14 5d 7 6s 2. It is the second densest elemental metal having an ambient pressure density of 22.65 g/cc at T 0 K and 22.56 g/cc at T 293. 
Iridium (atomic symbol: Ir, atomic number: 77) is a Block D, Group 9, Period 6 element with an atomic weight of 192.217. Thin Film Deposition & Evaporation Materials.Additive Manufacturing & 3D Printing Materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
